 As a result, there is a stronger attraction to the nucleus. The electrons are drawn in closer to the nucleus due to the strong positive charge on the nucleus, and the atom’s size shrinks. The number of protons and electrons increases over time as the atomic number increases, therefore the extra electrons are added to the same shell. In a period, the atomic size decreases from left to right. The valency of an element is determined by the number of electrons lost, gained, or shared by one atom to attain the nearest inert gas electron configuration. 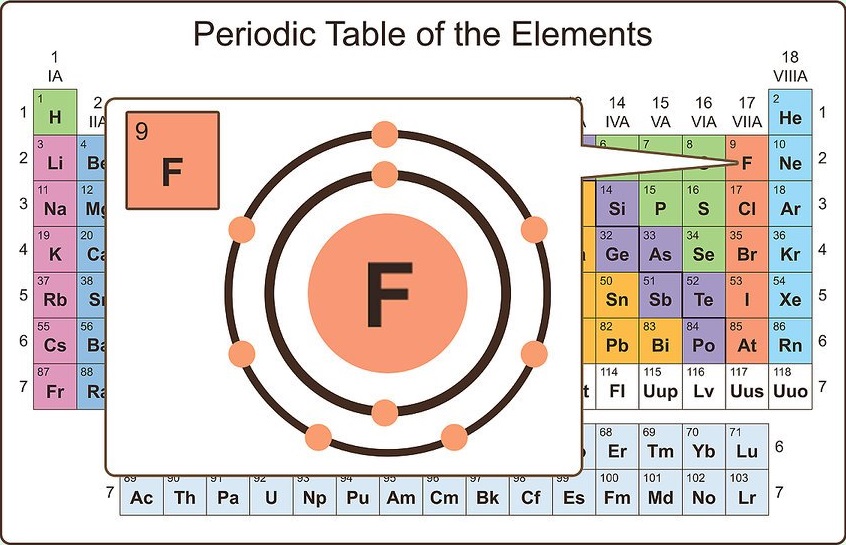
As a result, the valencies of elements from the same period differ. The valency increases from 1 in sodium to 4 in silicon in the third period of the periodic table, then decreases to zero in argon. Valency increases from 1 to 4 and then decreases to zero over a period from left to right. Sodium has a valency of 1, magnesium has a valency of 2, aluminium has a valency of 3, silicon has a valency of 4, phosphorous has a valency of 3, sulphur has a valency of 2, chlorine has a valency of 1, and argon has a valency of 0 in the third period. The valency of elements increases from 1 to 4 and eventually decreases to zero as you move from left to right in a period. For instance, elements of the third period, from sodium to argon, have atomic numbers ranging from 11 to 18. The atomic numbers of the elements in a period are also consecutive. Along the period, the number of valence electrons increases from 1 to 8. The number of electrons in the outermost shell of an atom grows from 1 to 8 as the electronic configurations of elements vary over time. Every period’s first element has one valence electron and every period’s last element has eight valence electrons, with the exception of the first period, where the last element, helium (He), has only two valence electrons. The element sodium (Na) contains one valence electron in the third period, whereas the element argon (Ar) has eight valence electrons. 
The number of valence electrons in elements increases from 1 to 8 as a period progresses from left to right, while it increases from 1 to 2 in the first period. These variations will be explained further below. Moving from left to right in a period of the periodic table, that is, moving from left to right in a horizontal row of the periodic table, we will discuss the variation of some of the important properties of elements including the number of valence electrons, valency, atom size, and metallic character. Group 1 is on the left side of the periodic table, whereas group 18 is on the right.Ĭharacteristics of Periods in a Periodic Table In the long form of the periodic table, there are 18 groups. The elements in a group do not have the same atomic number. 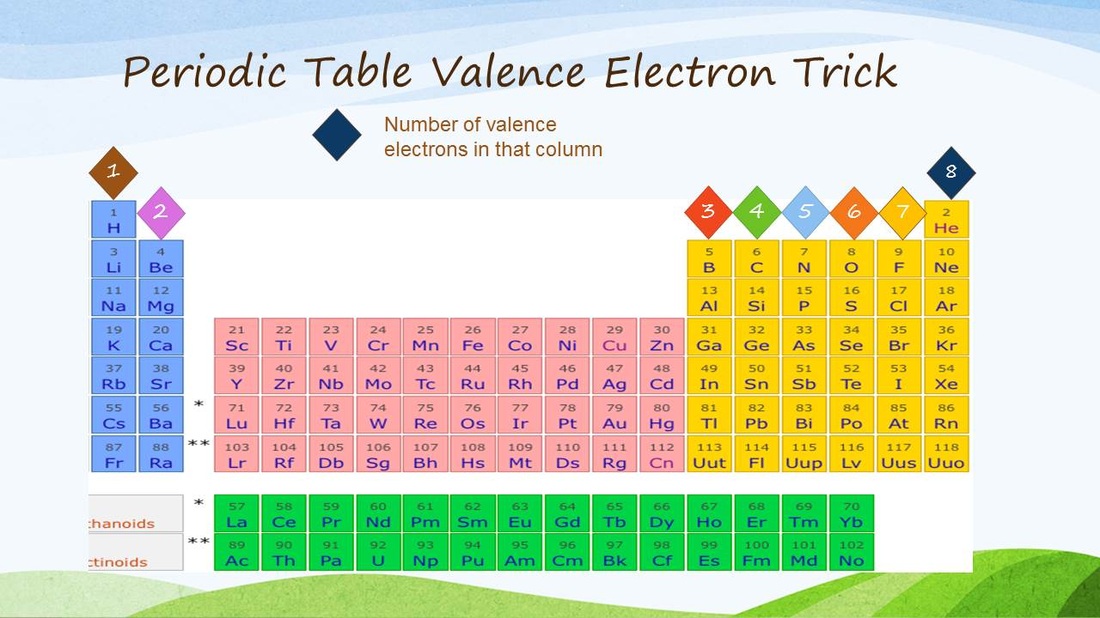
Groups– Groups are the vertical columns in a periodic table.The number of elements in a period is determined by the maximum number of electrons that may be accommodated in an atom’s various shells. The number of elements in each time varies. The atomic numbers of the elements in the periodic table are consecutive. There are seven periods in the periodic table. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
